Caesium and water plus a ph indicator11/9/2023  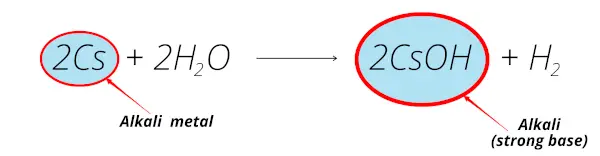
Further specimens have been cracked at 350Â☌ by a solution of CsI in a fused mixture of nitrates of rubidium, cesium, strontium and barium, by a similar mechanism. 
The specimens have been reexamined fractographically and metallographically in order to confirm that the cause of cracking was stress corrosion (SCC) and not delayed hydride cracking (DHC). Unirradiated split-ring specimens of Zircaloy fuel cladding, coated with CsI, cracked when stressed at elevated temperatures. Stress corrosion cracking of Zircaloys in unirradiated and irradiated CsI The results suggest that work function modification is a major contribution to the improved emission seen when CsI coatings are applied to C. It is demonstrated that the CsI coatings reduce the work function of the system significantly through a mechanism of induced dipoles. Therefore, an ab initio modeling investigation on the effects of thin CsI coatings on graphite has been carried out. Morgan, DaneĬesium- iodide (CsI)-coated graphite cathodes are promising electron sources for high power microwave generators, but the mechanism driving the improved emission is not well understood. Introducing the concept of the wettability of liquid chemical species of fission products in contact with solid fuels enabled developing accurate behavioural assessments of volatile fission products released by nuclear fuel.Īb initio study of the effects of thin CsI coatings on the work function of graphite cathodes Thus, volatile fission products released from molten nuclear fuels with complex internal composition and external structure migrate or evaporate to varying extents, depending on the nature of the solid-liquid interface and the fuel material surface, which becomes the pathway for the released fission products. We further observed the presence of CsI several tens of micrometres below the surface of the solid UO 2 sample, which would be caused by the infiltration of pores network by liquid CsI. We found experimentally that liquid CsI disperses extremely favourably toward solid UO 2, exhibiting a contact angle approaching zero. Certain fission-product behaviours remain unclear. Caesium and iodine caused environmental contamination and public exposure. In March 2011, the Fukushima Daiichi Nuclear Power Plant accident caused nuclear fuel to melt and the release of high-volatility fission products into the environment. Kurosaki, Ken Suzuki, Masanori Uno, Masayoshi Ishii, Hiroto Kumagai, Masaya Anada, Keito Murakami, Yukihiro Ohishi, Yuji Muta, Hiroaki Tanaka, Toshihiro Yamanaka, Shinsuke High wettability of liquid caesium iodine with solid uranium dioxide. CsOH still exists in steam atmosphere even at quite high temperature, up to around 1100 K. CsOH forms the most hydrated stable complexes followed by CsI, Csâ‚‚Iâ‚‚, and Cs. Interaction energies, standard enthalpies and Gibbs free energies of successive water molecules addition determine the ordering of the complexes. Thermodynamic properties obtained by these correlated ab initio calculations (entropies and thermal capacities at constant pressure as a function of temperature) are used in nuclear accident simulations using ASTEC/SOPHAEROS software. The second-order spin-free Douglas-Kroll-Hess Hamiltonian was used to account for the scalar relativistic effects. The calculations were performed using the coupled cluster theory including single, double and non-iterative triple substitutions (CCSD(T)) in conjunction with the basis sets (ANO-RCC) developed for scalar relativistic calculations. Structure and thermodynamic properties (standard enthalpies of formation and Gibbs free energies) of hydrated caesium species of nuclear safety interest, Cs, CsOH, CsI and its dimer Csâ‚‚Iâ‚‚, with one up to three water molecules, are calculated to assess their possible existence in severe accident occurring to a pressurized water reactor. Sudolská, Mária Cantrel, Laurent Cernušák, Ivan Microhydration of caesium compounds: Cs, CsOH, CsI and Csâ‚‚Iâ‚‚ complexes with one to three Hâ‚‚O molecules of nuclear safety interest. Although the preferred iodide is AgI, RbI and CuI additions also contribute to an improved polycrystalline CsI alloy with outstanding multispectral infrared transmittance properties. A transparent, strong CsI alloy is described having additions of monovalent iodides. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
